How does the antigen quick test work?
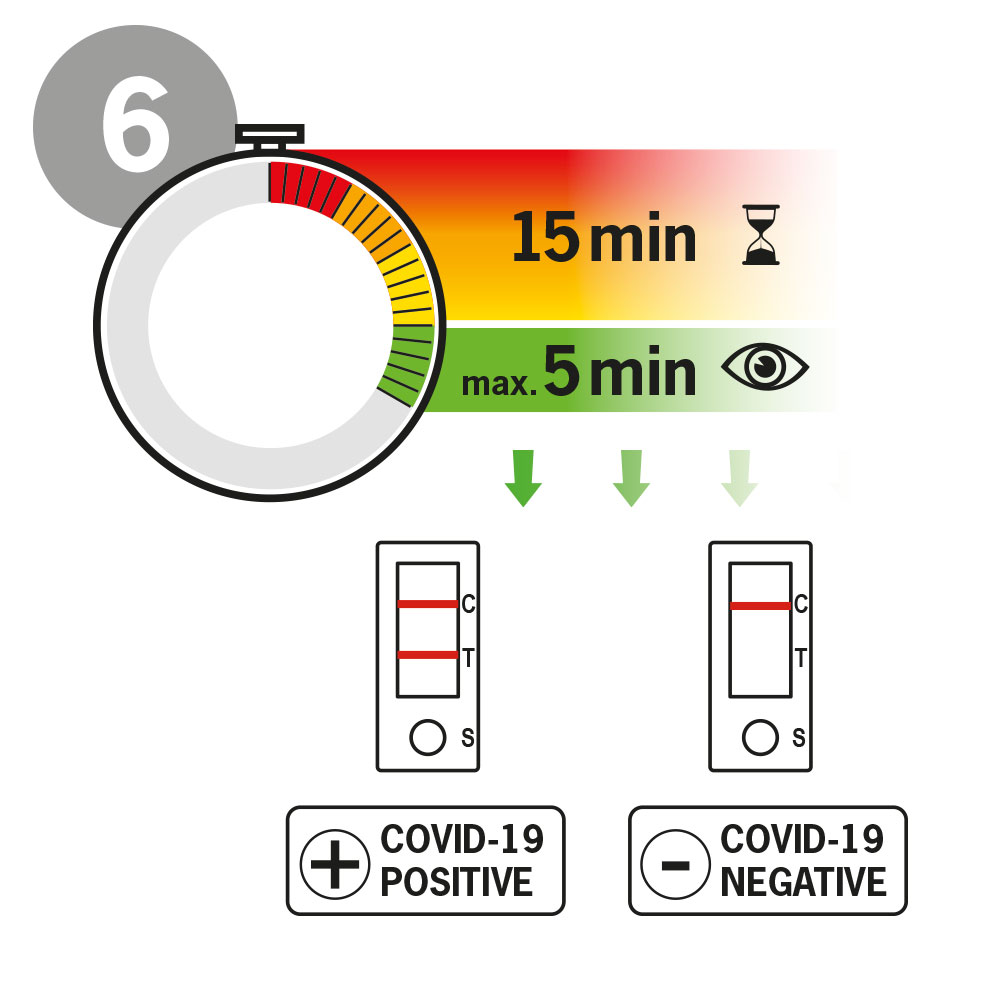
An antigen rapid test is a rapid test method that can be used to determine whether a person has an active Covid-19 infection. The test gives a positive or negative result after 15 minutes.
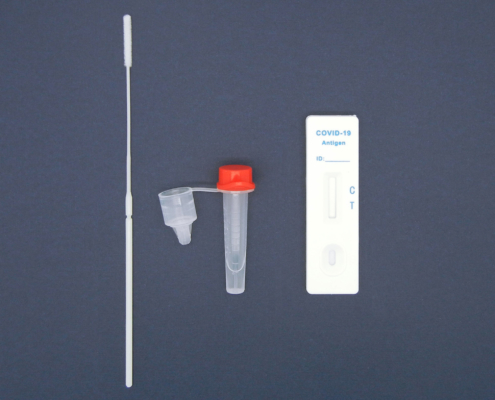
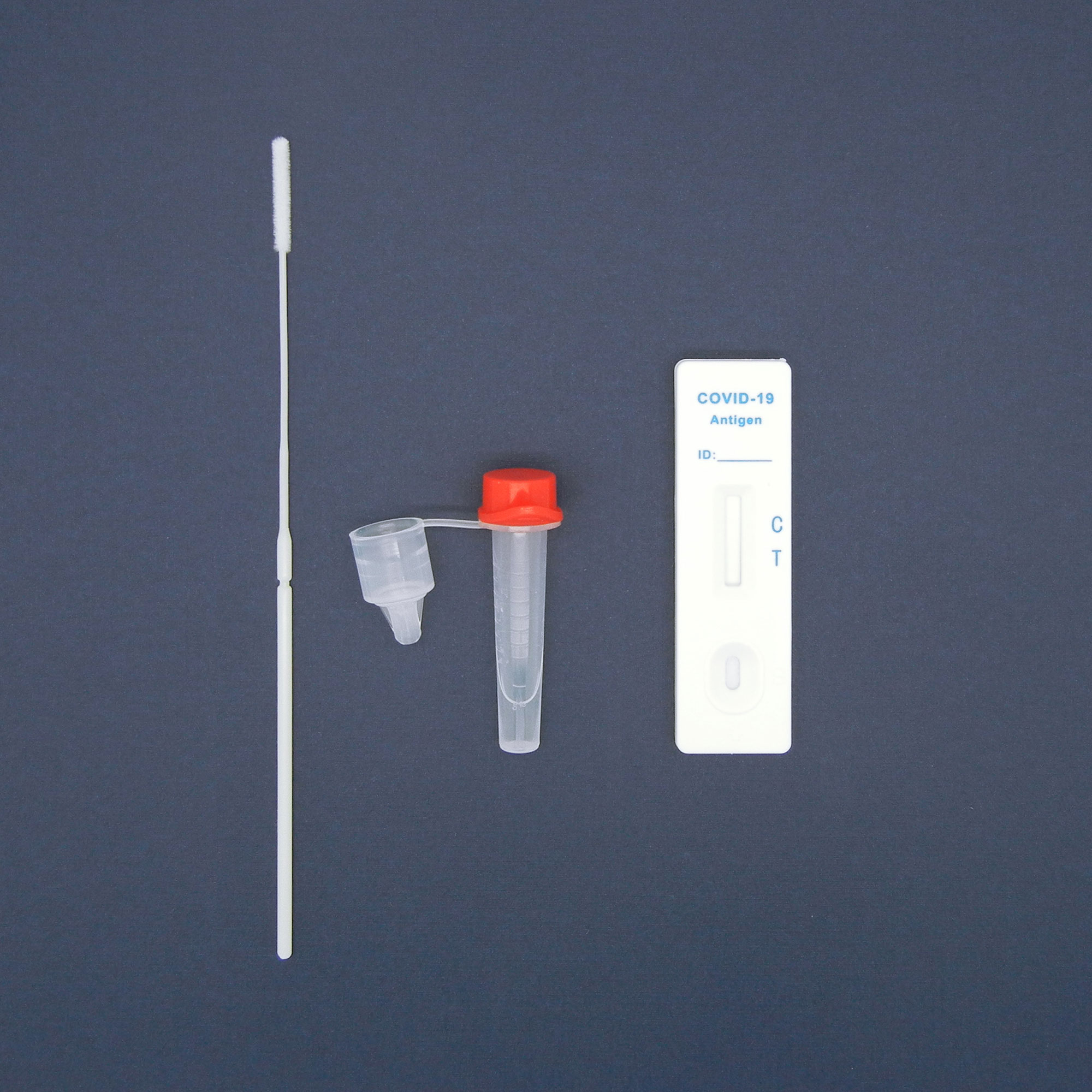
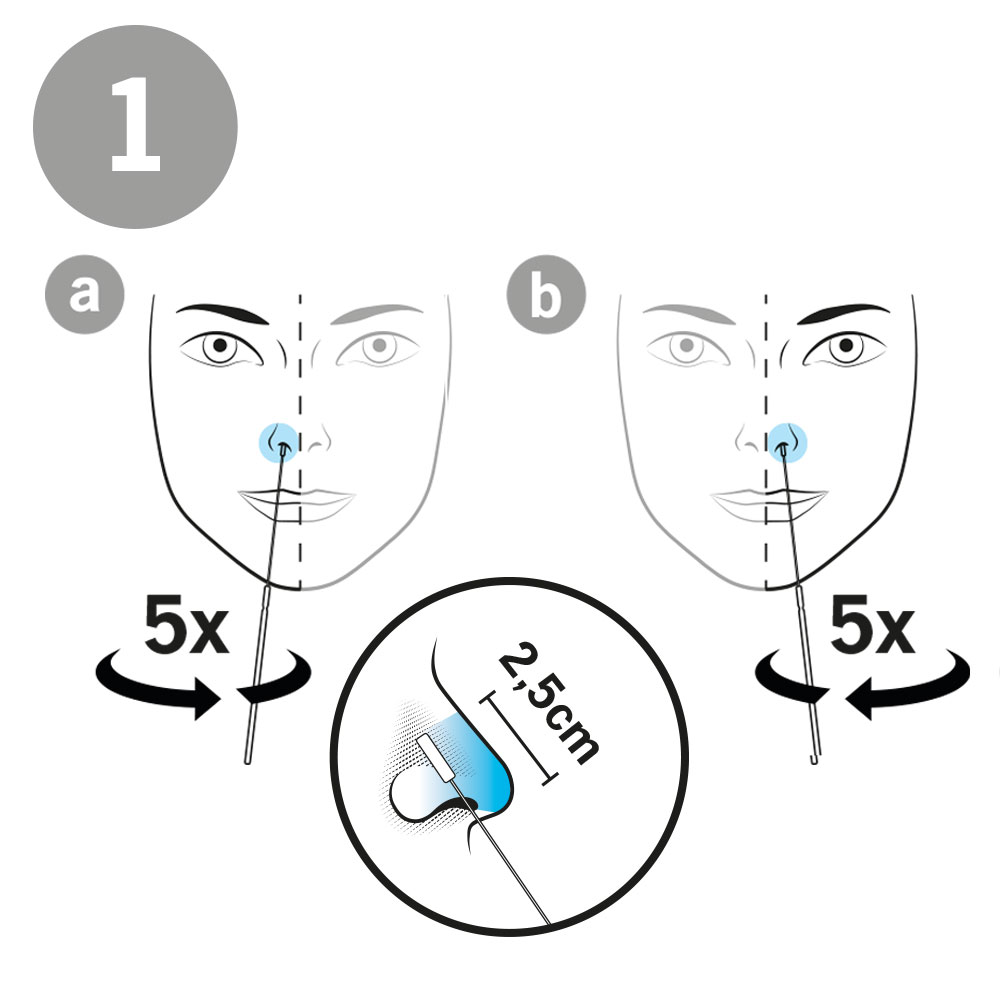
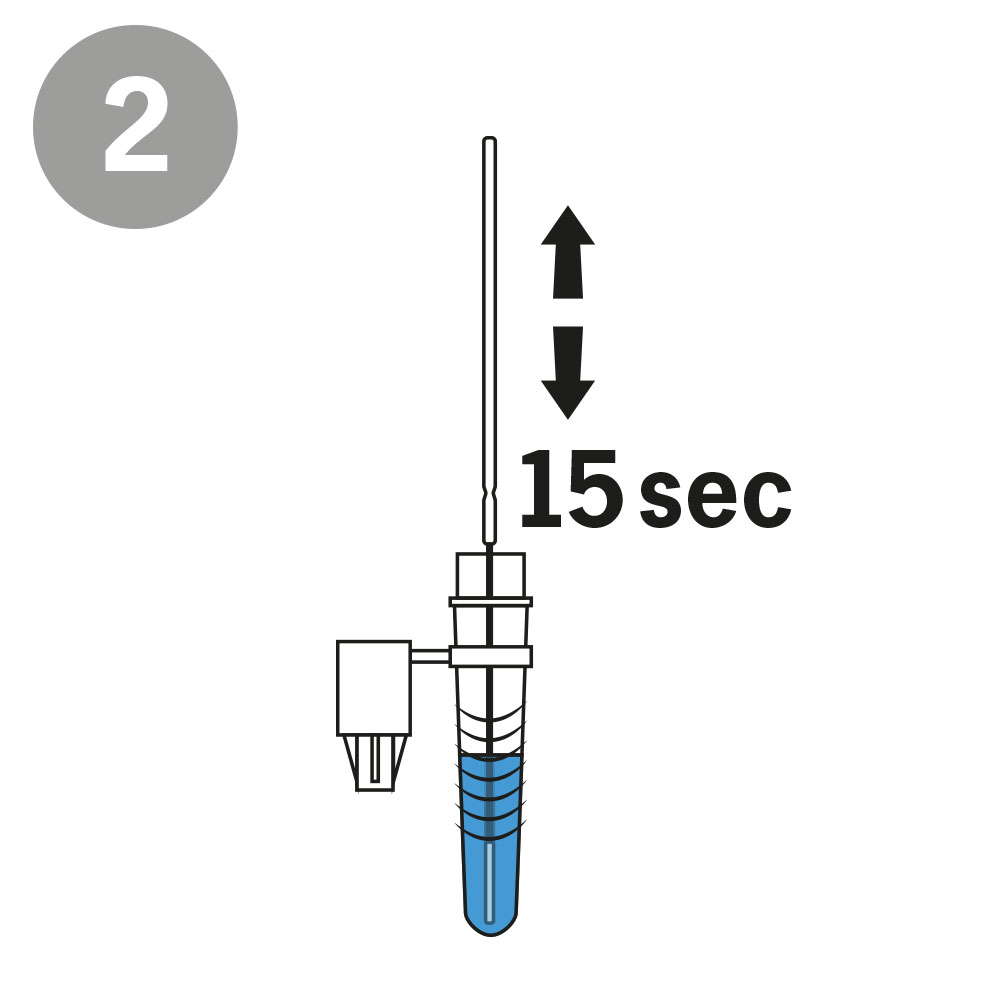
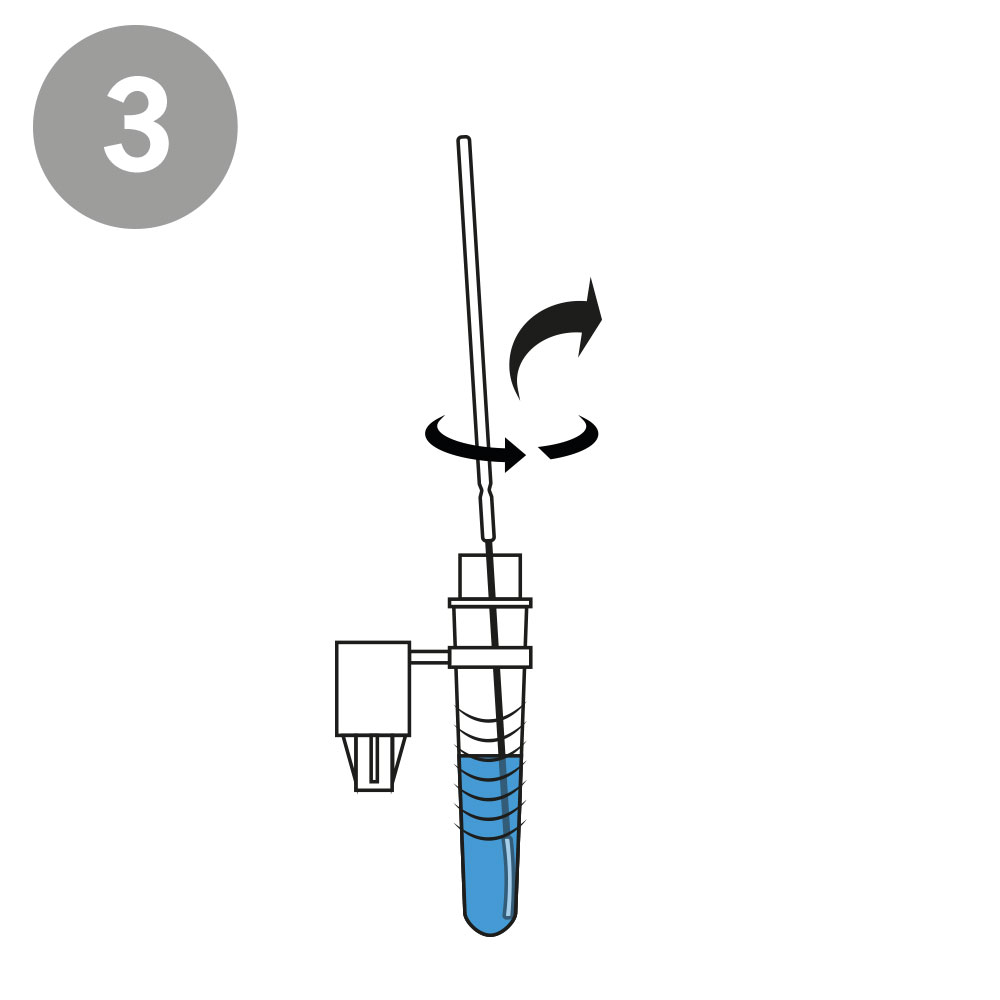
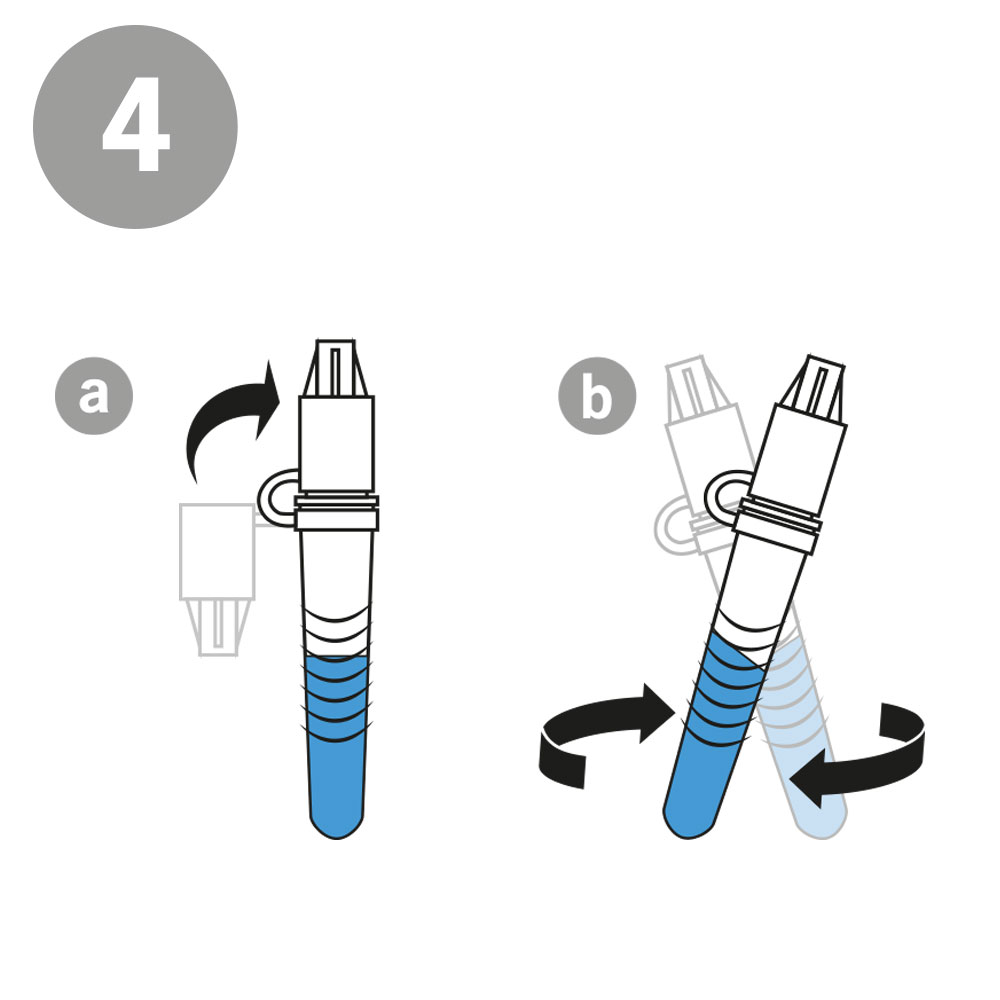
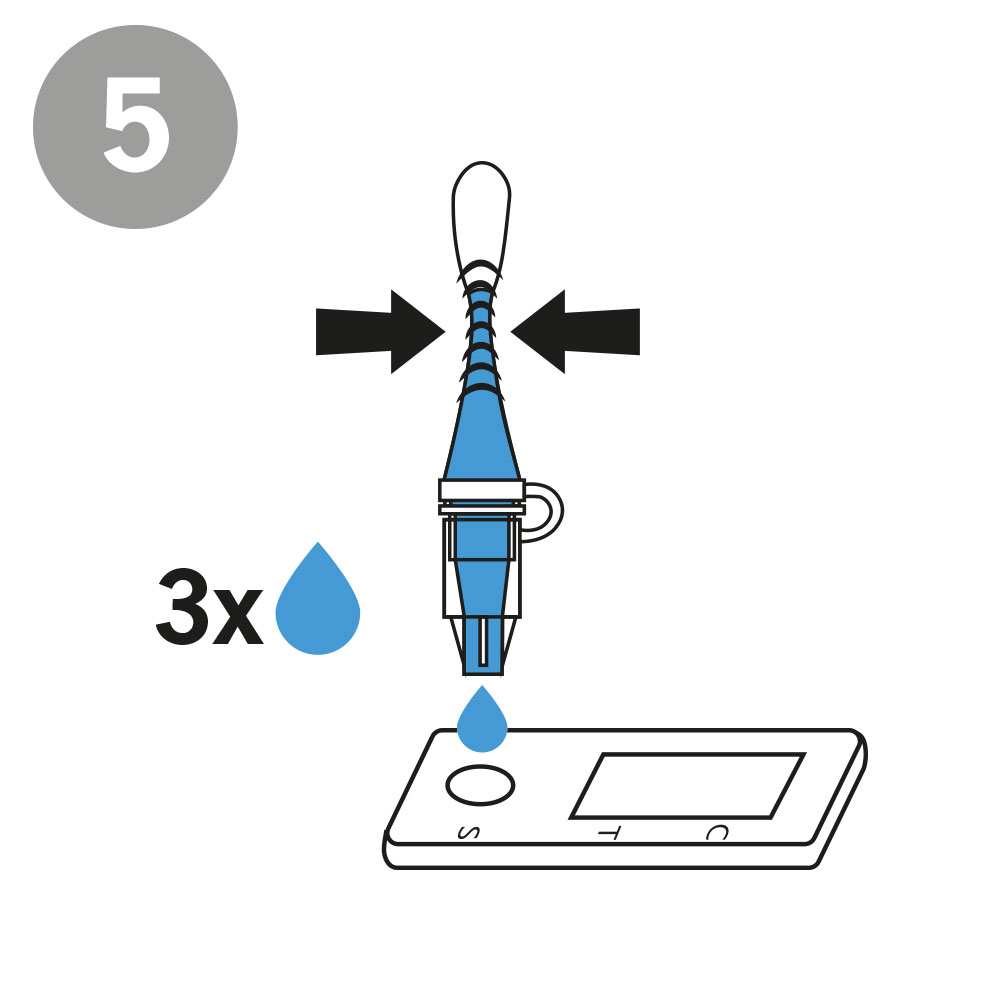
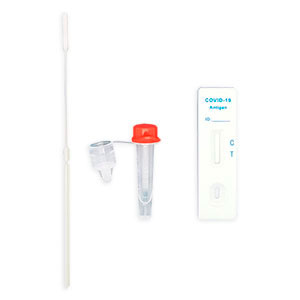
AESKU.RAPID quick test is performed by obtaining a swab in the nose. In this case, the test stick supplied is inserted into each nostril at a depth of 1.5 to 3 cm – in contrast to other tests, in which the stick must be inserted very deep into the nose. As the test can be considered minimally invasive it has been externally validated. The test also provides valid results when the swab is taken from the throat or nose-throat.
Since there are several rapid tests on the market with different quality and accuracy standards, it is not easy to recognize a safe, accurate and reliable test.
AESKU.RAPID quick test is CE-certified. It was developed by the established laboratory of the AESKU.GROUP in Wendelsheim, Germany and offers an excellent sensitivity of 100%. This makes it a reliable tool for combating the COVID-19 pandemic.
The test is reliable even with virus mutations. It uses the Nucleocapsid Protein Antigen and according to current data, its’ reliability is not affected by strain mutations.